
Xsensio raises USD 7 million to advance its wearable biosensing platform
3 March 2026
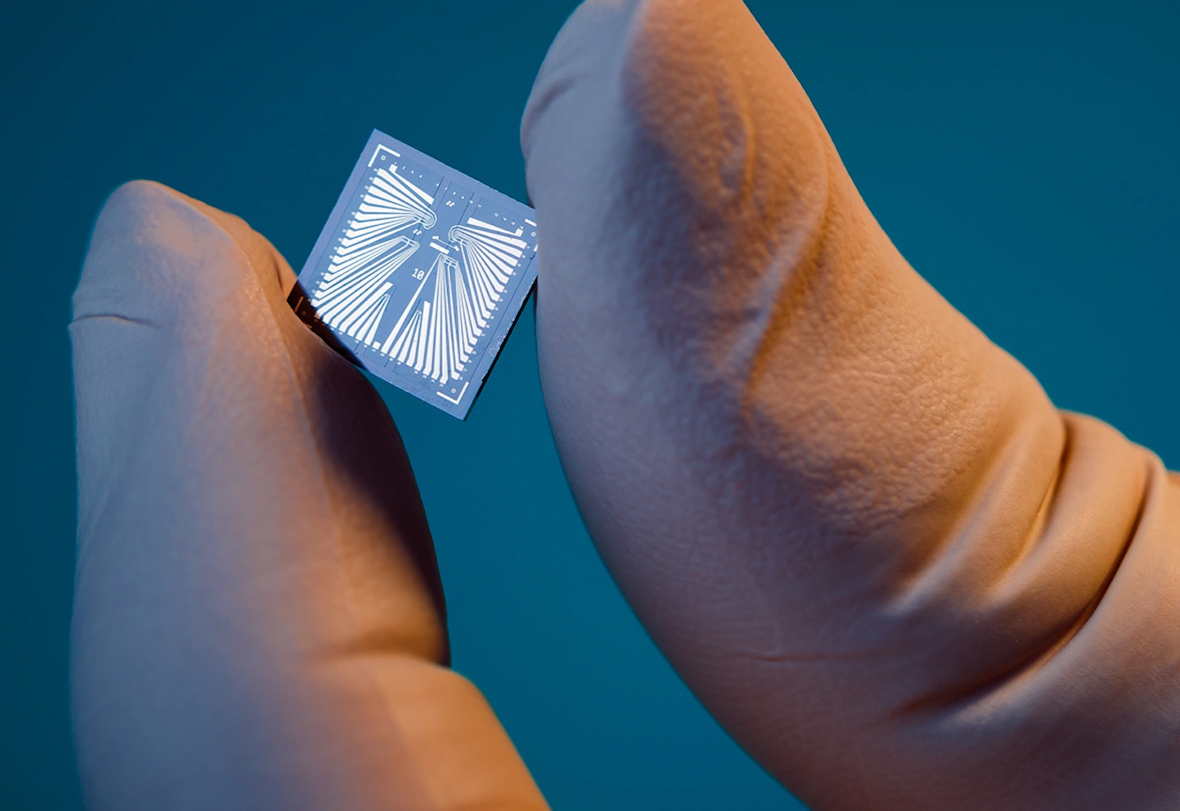 Xsensio’s Lab-on-Skin chip integrates multiple biosensors onto an ultra-miniaturized platform for continuous, real-time biochemical monitoring. | © Xsensio
Xsensio’s Lab-on-Skin chip integrates multiple biosensors onto an ultra-miniaturized platform for continuous, real-time biochemical monitoring. | © Xsensio
Lausanne-based Xsensio has closed an oversubscribed USD 7 million Series A financing round to accelerate clinical deployment of its Lab-on-Skin wearable biosensing platform, with new backing from WI Harper and a collaboration with Texas Instruments.
Lausanne-based deep-tech company Xsensio has successfully closed a USD 7 million oversubscribed Series A financing round to accelerate the clinical validation and deployment of its Lab-on-Skin wearable biosensing platform. The round was led by San Francisco-based venture capital firm WI Harper, with participation from Privilège Ventures, the European Innovation Council, and private investors across the United States, Europe, and Asia.
Developed at the EPFL Innovation Park, the Lab-on-Skin platform integrates multiple sensors onto an ultra-miniaturized chip capable of continuously monitoring a range of biomarkers, from metabolites and ions to proteins and hormones, through interstitial fluid on the skin. Its modular architecture allows the same platform to be adapted across diverse clinical applications, supporting real-time biochemical insight at the point of care.
Alongside the financing, Xsensio announced a long-term collaboration with Texas Instruments, a global semiconductor leader. The partnership targets CMOS integration, miniaturization, and large-scale manufacturability, strengthening the platform’s path toward scalable industrial deployment.
The Series A builds on Xsensio’s earlier partnership with Mayo Clinic, announced in 2023, which focused on clinical validation for critical care monitoring in hospital and home settings. The new funding advances the company’s next phase: translating continuous biochemical monitoring into live clinical environments.

